Research Area
- Multi-Functional Nano-Sciences of Advanced Metal Complexes
Research Subjects
- Strongly Electron Correlated Nanowire Metal Complexes
- Single Chain Magnets, Single Molecule Magnets
- Orgainc-Lanthanide Hybrid Luminescent Complexes
- Materials Science of Bistable State in the Condensed Solid
- Inorganic-Organic Hybrid Superconductor
Overview of Research
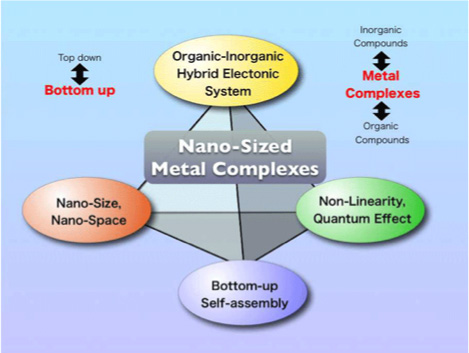
Recently, nano-sciences or nano-technologies have been attracting much attention because they show very interesting physical properties based on the non-linearity and quantum effect. There are two methods to obtain the nano-size materials, that is, "top-down" and "bottom-up" methods. The top-down method such as laser abrasion has a limitation to make particles with the sizes less than 100 nm. On the other hand, the bottom-up method is promising to control the nano-sizes since the chemical reactions are available. However, the researchers based on the bottom-up methods are not so many and such methods have not been accomplished so far. Then, we would like to focus on the bottom-up methods. As for the bottom-up methods, there are three types of the target materials such as inorganic compounds, organic compounds, and metal complexes. The inorganic compounds easily take 3-dimensional bulk structures. The organic compounds easily take 0- and 1-dimensional bulk materials. Therefore, neither inorganic nor organic compounds are suitable for the nano-sciences. On the other hand, the metal complexes easily take nano-size clusters where they are surrounded with the organic ligands. Therefore, the nano-sciences of the advanced metal complexes are more promising. The nano-sciences of advanced metal complexes are composed of four key terms as follows: 1)Organic-inorganic hybrid electronic systems, 2)Nano-sizes or nano-spaces, 3)Bottom-up or self-assembly methods, and 4)Non-linearity or quantum effect. In the field of nano-sciences of advanced metal complexes, we will focus on the multi-functionalities such as a bi-functionality between optics and conductivity, a bi-functionality between conductivity and magnetism, and a bi-functionality between magnetism and optics. These are for example, the photo-induced switching between single-chain quantum magnets and paramagnets, the conducting single-molecule quantum magnets or single-chain quantum magnets, and the conducting optical nonlinearity. These are perfectly new scientific fields.
Selected Publications
- Comparison of the Magnetic Anisotropy and Spin Relaxation Phenomenon of Dinuclear Terbium(III) Phthalocyaninato Single-Molecule Magnets Using the Geometric Spin Arrangement
T. Morita, M. Damjanović, K. Katoh, Y. Kitagawa, N. Yasuda, Y. Lan, W.Wernsdorfer, B. K. Breedlove, M. Enders, and M. Yamashita
J. Am. Chem. Soc., 140, 2995-3007 (2018).
- Multiple Hydrogen-Bond Approach to Uncommon Pd(III) Oxidation State: A Pd-Br Chain with High Conductivity and Thermal Stability
M. R. Mian, H. Iguchi, S. Takaishi, H. Murasugi, T. Miyamoto, H. Okamoto, H. Tanaka, S. Kuroda, B. K. Breedlove and M. Yamashita
J. Am. Chem. Soc., 139, 6562-6565 (2017).
- Direct Observation of Ordered [4HS-2LS] and [2HS-4LS] Intermediate States of an Iron(lll) Three-Step Spin-Crossover Complex
Z. Y. Li, H. Ohtsu, T. Kojima, J. W. Dai, T. Yoshida, B. K. Breedlove, W. X. Zhang, H. Iguchi, O. Sato, M. Kawano and M. Yamashita
Angew. Chem. Int. Ed., 55, 1-7 (2016).
- First Observation of a Kondo Resonance for a Stable Neutral Pure Organic Radical, 1,3,5-Triphenyl-6-Oxoverdazyl, Adsorbed on Au(III).
J. Liu, H. Issiki, K.Katoh, T. Morita, B. K. Breedlove, M. Yamashita, and T. Komeda
J. Am. Chem. Soc. 135, 651-658 (2013).
- New Approach for Designing Single-Chain Magnets: Organization Of Chains via Hydrogen Bonding between Nucleobases.
W. -X. Zhang, T. Shiga, H. Miyasaka, and M. Yamashita
J. Am. Chem. Soc. 134, 6908-6911 (2012).
- Observation and electric current control of a local spin in a single-molecule magnet.
T. Komeda, H. Isshiki, J. Liu, Y. F. Zhang, N. Lorente, K. Katoh, B. K. Breedlove, and M. Yamashita
Nature Commun. 2, 217-223 (2011).
- Stabilization of Pd(III) States in Nano-Wire Coordination Complexes.
M. Yamashita and S. Takaishi
Chem. Commun. (Feature Article) 46, 4438-4448 (2010).
- Electroconductive Porous Coordination Polymer Cu[Cu(pdt)2] Composed of Doner and Acceptor Building Units.
S. Takaishi, M. Hosoda, T. Kajiwara, H. Miyasaka, M. Yamashita, Y. Nakanishi, Y. Kitagawa, K. Yamaguchi, A. Kobayashi, and H. Kitagawa
Inorg. Chem. 48, 9048-9050 (2009).
- Direct Observation of Lanthanide(III)-Phthalocyanine Molecules on Au(111) by Using Scanning Tunneling Microscopy and Scanning Tunneling Spectroscopy and Thin-Film Field-Effect Transistor Properties of Tb(III) and Dy(III)-Phthalocyanine Molecules.
K. Katoh, Y. Yoshida, M. Yamashita, H. Miyasaka, B. K. Breedlove, T. Kajiwara, S. Takaishi, N. Ishikawa, H. Isshiki, Y. Zhang, T. Komeda, M. Yamaguchi, and J. Takeya
J. Am. Chem. Soc. 131, 9967-9976 (2009).
- Charge-Density-Wave to Mott-Hubbard Phase Transition in Quasi-One-Dimensional Bromo-Bridged Pd Compounds.
S. Takaishi, M. Takamura, T. Kajiwara, H. Miyasaka, M. Yamashita, M. Iwata, H. Matsuzaki, H. Okamoto, H. Tanaka, S. Kuroda, H. Nishikawa, H. Oshio, K. Kato, and M. Takata
J. Am. Chem. Soc. 130, 12080-12084 (2008).
Contact Information
“Please delete “/” after “@” in a mail address.”
Shinya TAKAISHI
(TEL +81-22-795-6545, Email: s-takaishi@/mail.tains.tohoku.ac.jp)
Website: http://web.tohoku.ac.jp/sakutai/