English version is available at the bottom of this page.
生物物理工学
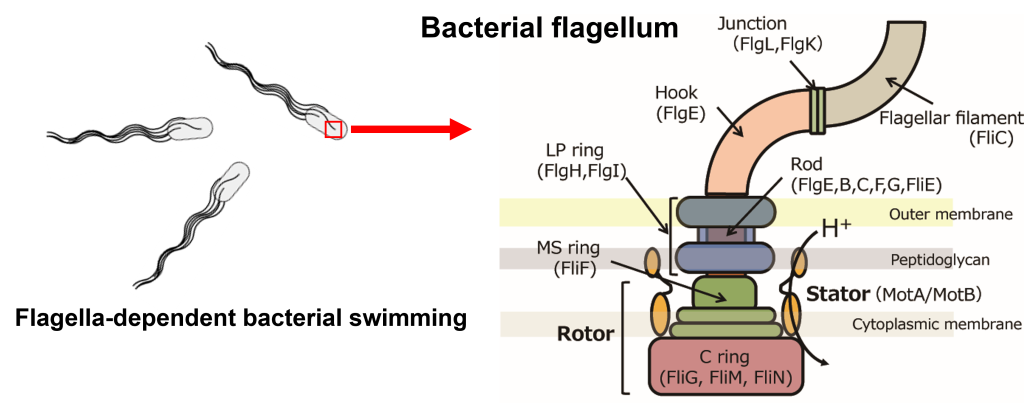
1.生体分子モーターのエネルギー変換機構の解明と応用
粘性と熱揺らぎが支配する微小な流体環境(低レイノルズ数領域)において,バクテリア等の微生物がどのように効率的な運動を実現しているのか、その物理的原理を解明します。主にバクテリア運動を司る「べん毛」という直径45ナノメートルの回転分子モーターを題材に,高精度なイメージング技術を駆使してミクロスケールのエネルギー変換機構を解明します。得られた知見を,マイクロマシンの設計やドラッグデリバリーシステムの開発へと展開することを目指します。
関連する成果
Nakamura et al. (2010) PNAS “Evidence for symmetry in the elementary process of bidirectional torque generation by the bacterial flagellar motor”
Tahara et al. (2018) Sci. Adv. “The mechanism of two-phase motility in the spirochete Leptospira: Swimming and crawling”
Suzuki et al. (2019) J. Bacteriol. “Effect of the MotA(M206I) mutation on torque generation and stator assembly in the Salmonella H+-driven flagellar motor”
Nakamura & Minamino (2024) Biomolecules “Structure and dynamics of the bacterial flagellar motor complex” [Review article] [Editor’s Choice]
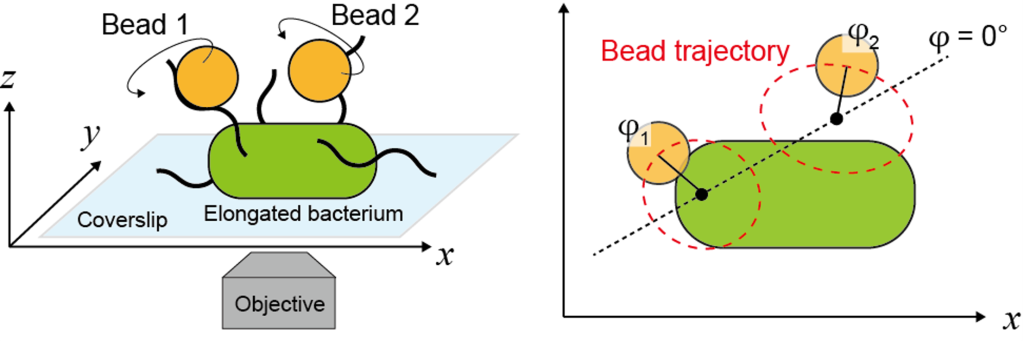
2.低レイノルズ数領域の同期現象
生命システムにおけるリズムと同期は,生命現象の根幹をなす重要な要素です。本テーマでは,微生物の運動や繊毛の動きに見られる「流体力学的同期」に着目します。非線形物理学の枠組みと高精度な一分子イメージングを組み合わせ,ミクロな流体相互作用がどのようにアクティブ振動子間の秩序ある動きを生み出すのかを探究します。我々は,近接したバクテリアのべん毛が断続的に同位相同期することを実証し,流体力学モデルともよく一致することを示しました。実験と理論の両面から,生命現象特有の同期現象の背後に潜む物理法則の解明に挑みます。
関連する成果
Ishihara et al. (2026) bioRxiv “Emergent hydrodynamic synchronization between microbeads labeling bacterial flagellar motors”
3.ナノ・マイクロスケールの計測と制御
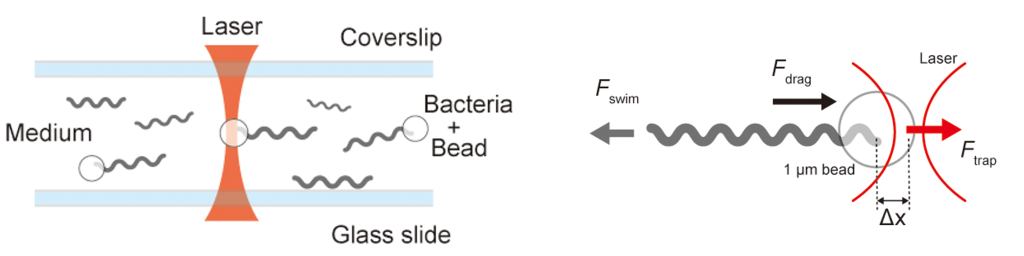
光ピンセット法は,集光した赤外レーザーで微小物体をまさにピンセットのようにつまむ(トラップする)技術です。物理的な摂動に対する生命システムの応答を定量化するこの技術は,微小空間における制御・計測法として広く利用されています。赤外レーザーでトラップした生体試料を任意に操作して局所的な流体力を印加し,その応答を解析することで,細胞や生体材料の機械的強度や微視的な物性を明らかにします。たとえば,バクテリア1細胞の遊泳力や細胞剛性の定量評価が可能です。
関連する成果
Abe et al. (2020) Sci. Rep. “Implications of back-and-forth motion and powerful propulsion for spirochetal invasion”
Abe, Koizumi, Takazaki et al. (2026) bioRxiv “A novel periplasmic layer formed by an outer membrane lipo-protein governs the cell-envelope integrity and stiffness of Leptospira interrogans“
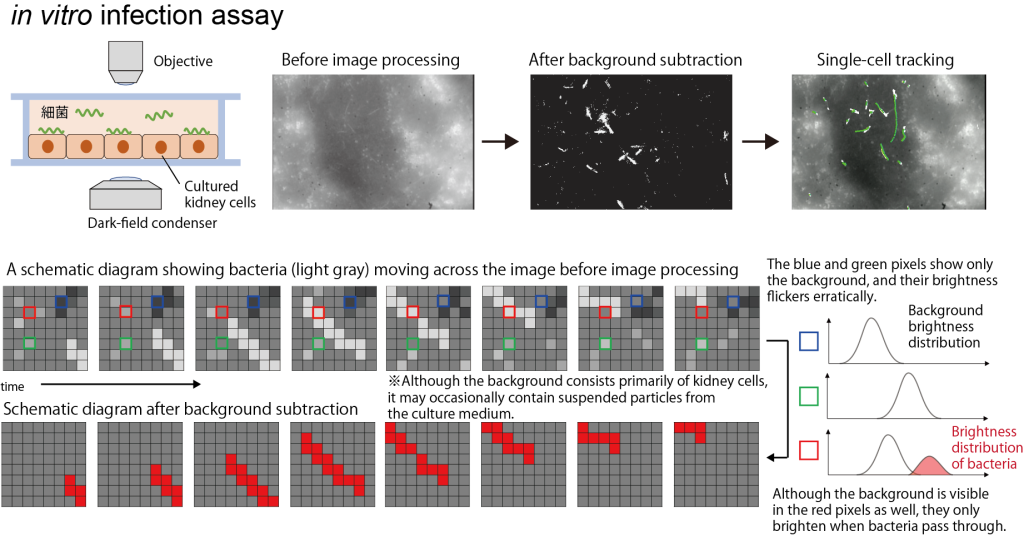
4.AI画像解析による感染症研究
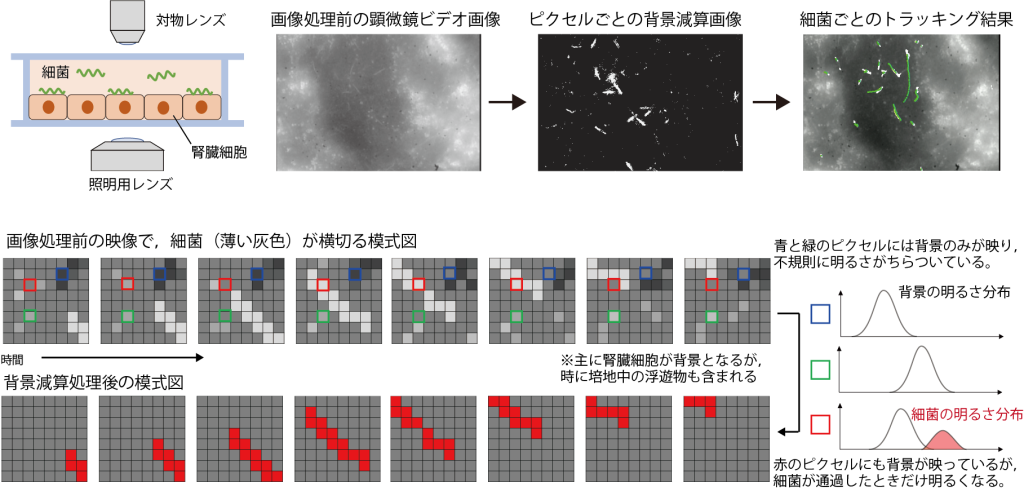
AIを用いた画像解析(コンピュータビジョン)は,情報や物理・工学の分野だけでなく,生命科学の基礎研究においても不可欠な技術となりつつあります。例えば,ベイズの定理に基づいて「動く」前景を「動かない」背景と区別する背景差分法といった技術を用いると,培養細胞に感染させた病原菌を無標識で検出することができます。データサイエンスと生命科学の架け橋となり,感染機構の解明や,迅速な診断・創薬支援につながる基盤技術を構築します。
関連する成果
Abe et al. (2023) Nat. Commun. “Machine learning-based motion tracking reveals an inverse correlation between adhesivity and surface motility of the leptospirosis spirochete”
Biophysics and Bioengineering
-300x258.png)
1. Elucidation and application of energy conversion mechanisms in biomolecular motors
We aim to elucidate the physical principles of how microorganisms, such as bacteria, achieve efficient motility in microscopic fluid environments dominated by viscosity and thermal fluctuations (low Reynolds number regime). Focusing on the bacterial flagellum—a rotary molecular motor only 45 nanometers in diameter—we utilize high-precision imaging techniques to uncover the energy conversion mechanisms at the microscale. Our goal is to apply these insights to the design of micromachines and the development of drug delivery systems.
Selected publications
Nakamura et al. (2010) PNAS “Evidence for symmetry in the elementary process of bidirectional torque generation by the bacterial flagellar motor”
Tahara et al. (2018) Sci. Adv. “The mechanism of two-phase motility in the spirochete Leptospira: Swimming and crawling”
Suzuki et al. (2019) J. Bacteriol. “Effect of the MotA(M206I) mutation on torque generation and stator assembly in the Salmonella H+-driven flagellar motor”
Nakamura & Minamino (2024) Biomolecules “Structure and dynamics of the bacterial flagellar motor complex” [Review article] [Editor’s Choice]

2. Synchronization phenomena in low Reynolds number regimes
Rhythm and synchronization are fundamental elements of biological systems. This research theme focuses on “hydrodynamic synchronization” observed in microbial motility and ciliary motion. By combining nonlinear physics frameworks with high-precision single-molecule imaging, we explore how microscopic fluid interactions generate ordered motion among active oscillators. We have demonstrated that the flagella of adjacent bacteria synchronize in-phase intermittently, a finding that aligns well with hydrodynamic models. We challenge ourselves to uncover the underlying physical laws of bio-specific synchronization through both experimental and theoretical approaches.
Selected publications
Ishihara et al. (2026) bioRxiv “Emergent hydrodynamic synchronization between microbeads labeling bacterial flagellar motors”

3. Nano- and microscale measurement and control
Optical tweezers are a technology that uses focused infrared lasers to “trap” and manipulate microscopic objects just like physical tweezers. This technique is widely used for measurement and control in micro-spaces, allowing us to quantify the response of biological systems to physical perturbations. By manipulating trapped biological samples and applying localized fluid forces, we analyze their responses to reveal the mechanical strength and microscopic physical properties of cells and biomaterials. For instance, this allows for the quantitative evaluation of the swimming force of a single bacterium or cell stiffness.
Selected publications
Abe et al. (2020) Sci. Rep. “Implications of back-and-forth motion and powerful propulsion for spirochetal invasion”
Abe, Koizumi, Takazaki et al. (2026) bioRxiv “A novel periplasmic layer formed by an outer membrane lipo-protein governs the cell-envelope integrity and stiffness of Leptospira interrogans“

4. Infectious disease research via AI-based image analysis
AI-driven image analysis (computer vision) is becoming an indispensable technology not only in information science, physics, and engineering but also in fundamental life science research. For example, by using techniques such as “background subtraction”—which distinguishes a “moving” foreground from a “stationary” background based on Bayes’ theorem—we can detect pathogens infecting cultured cells without the need for labeling. We aim to bridge data science and life sciences to build foundational technologies that lead to the elucidation of infection mechanisms, rapid diagnostics, and support for drug discovery.
Selected publications
Abe et al. (2023) Nat. Commun. “Machine learning-based motion tracking reveals an inverse correlation between adhesivity and surface motility of the leptospirosis spirochete”